
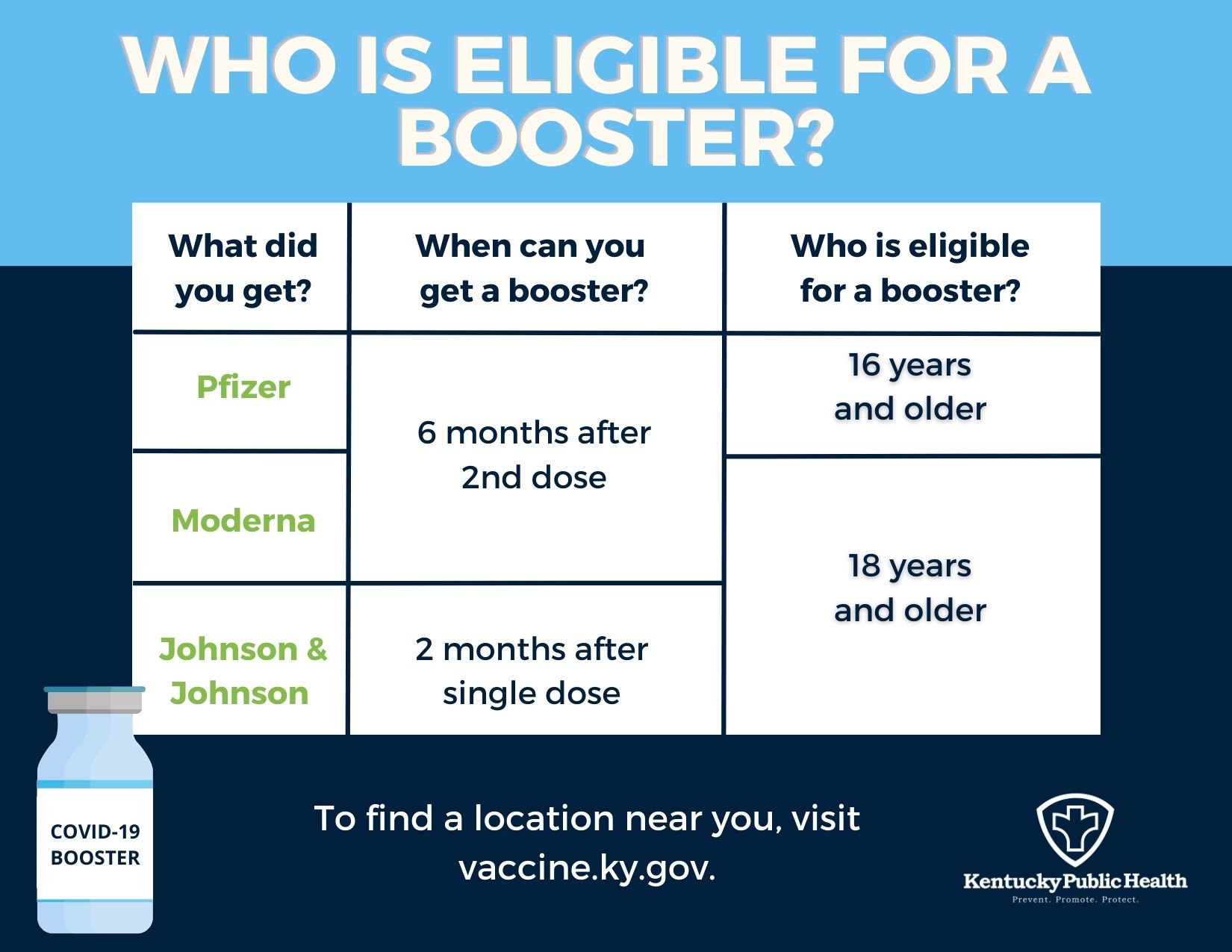
Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

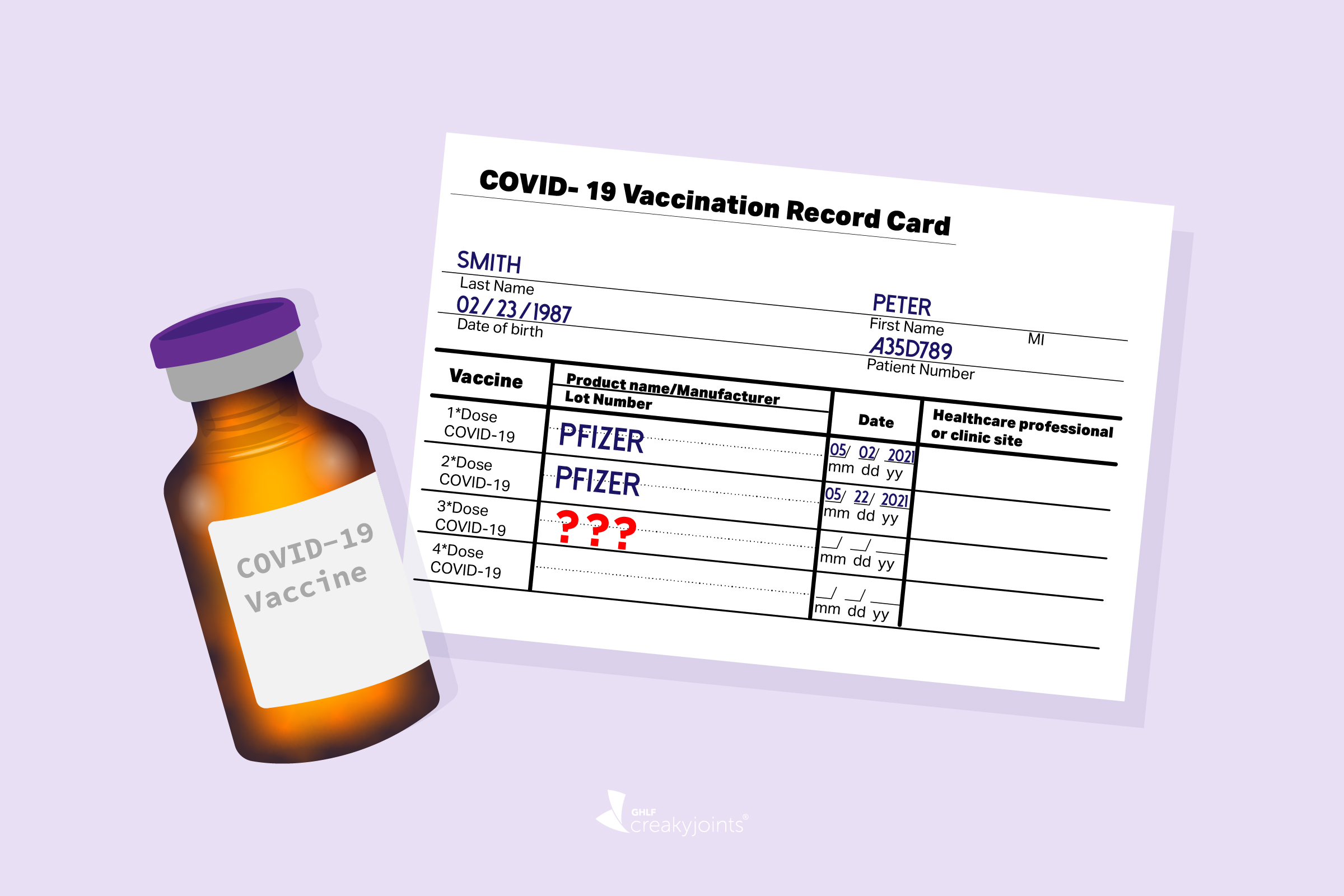
CDC on X: "CDC recommends that certain populations receive a booster shot of Pfizer-BioNTech's #COVID19 Vaccine at least 6 months after completion of their 2-dose Pfizer-BioNTech vaccine series. See full details: https://t.co/77CTFv1g4m.










.png)

