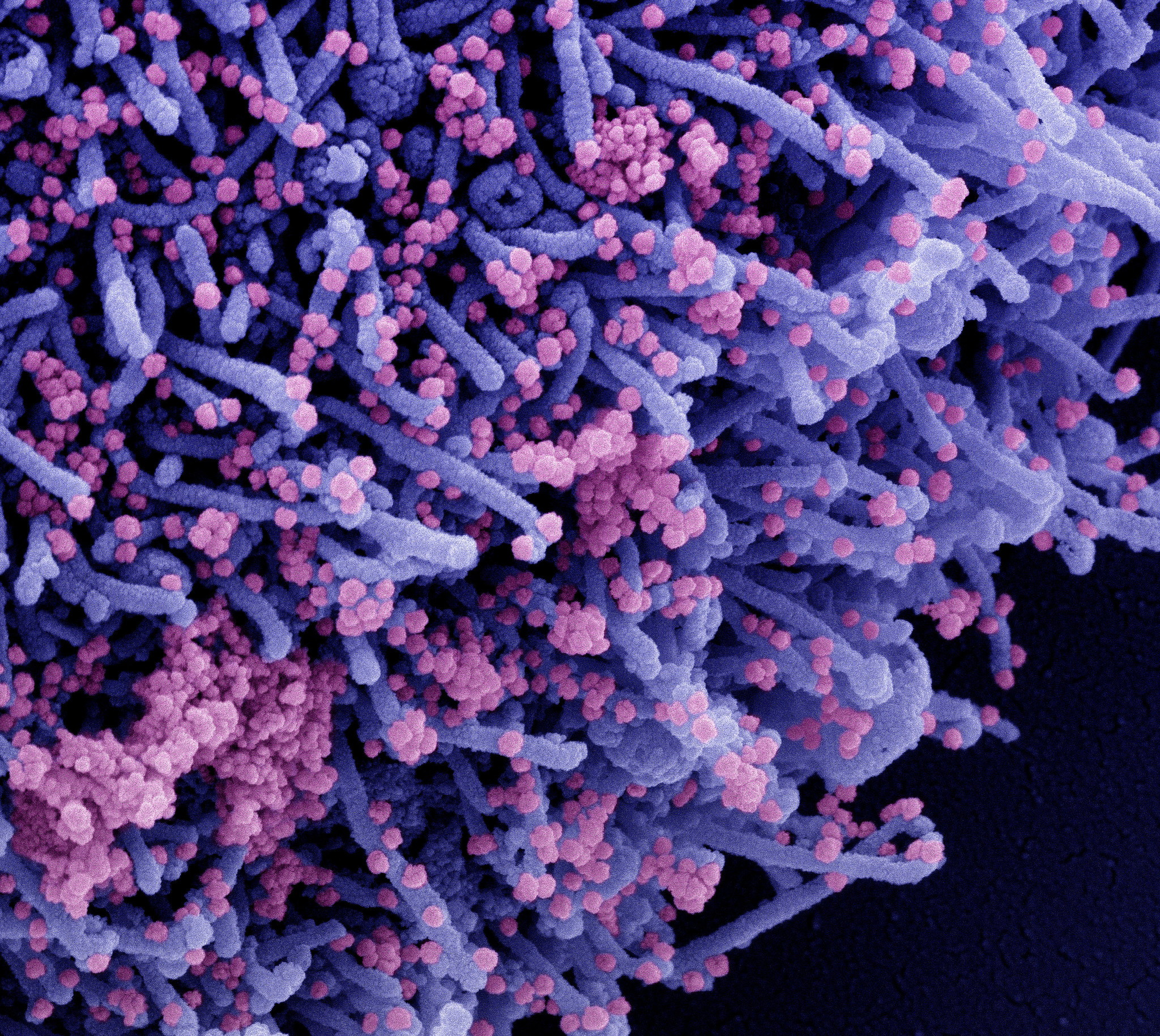
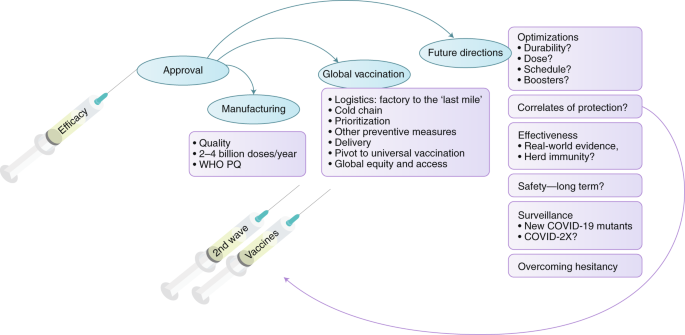
Immunogenicity and safety of NVSI-06-07 as a heterologous booster after priming with BBIBP-CorV: a phase 2 trial | Signal Transduction and Targeted Therapy

Immunogenicity and reactogenicity of BNT162b2 booster in ChAdOx1-S-primed participants (CombiVacS): a multicentre, open-label, randomised, controlled, phase 2 trial - The Lancet

Safety and immunogenicity of a bivalent SARS-CoV-2 protein booster vaccine, SCTV01C, in adults previously vaccinated with mRNA vaccine: a randomized, double-blind, placebo-controlled phase 1/2 clinical trial - eBioMedicine

Protocol for a multicentre randomised controlled trial examining the effects of temporarily pausing Bruton tyrosine kinase inhibitor therapy to coincide with SARS-CoV-2 vaccination and its impact on immune responses in patients with

Effect of a 2-week interruption in methotrexate treatment versus continued treatment on COVID-19 booster vaccine immunity in adults with inflammatory conditions (VROOM study): a randomised, open label, superiority trial - The Lancet

Department of Health and Social Care on Twitter: "Everyone aged 16 and over is eligible for the #COVID19 booster from three months after their second dose. Book an appointment online or find
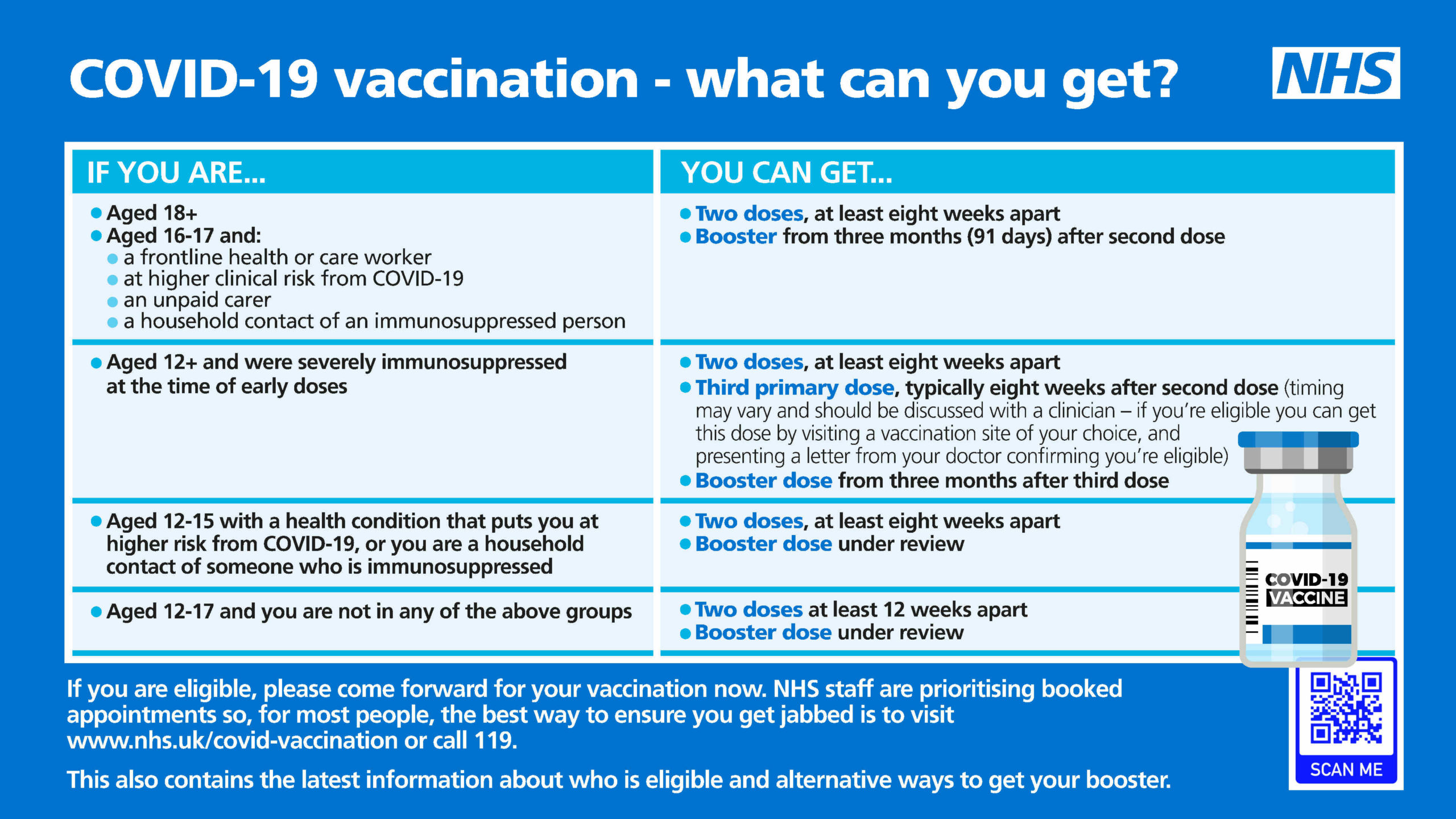
COVID vaccinations: questions and answers about the rollout in North Yorkshire - NHS North Yorkshire CCG
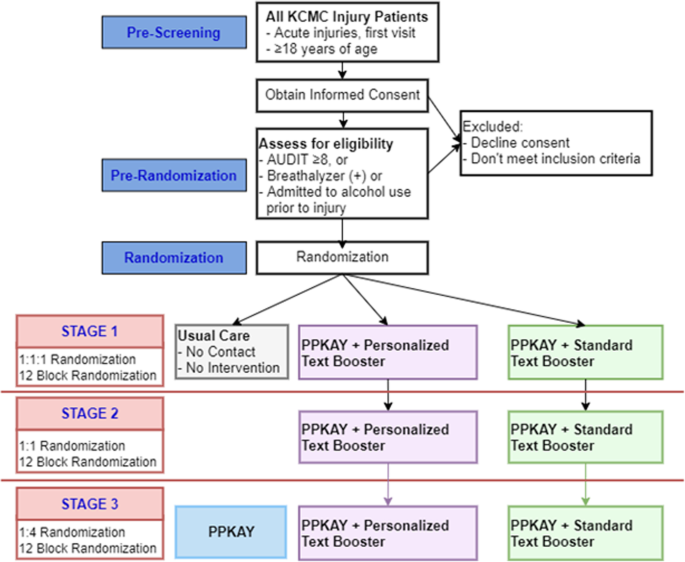
PRACT: a pragmatic randomized adaptive clinical trial protocol to investigate a culturally adapted brief negotiational intervention for alcohol use in the emergency department in Tanzania | Trials | Full Text

Effects of temporarily suspending low-dose methotrexate treatment for 2 weeks after SARS-CoV-2 vaccine booster on vaccine response in immunosuppressed adults with inflammatory conditions: protocol for a multicentre randomised controlled trial and ...

Quadrivalent meningococcal tetanus toxoid-conjugate booster vaccination in adolescents and adults: phase III randomized study | Pediatric Research

Pneumococcal conjugate vaccine 13 delivered as one primary and one booster dose (1 + 1) compared with two primary doses and a booster (2 + 1) in UK infants: a multicentre, parallel

Immunogenicity of a single-dose compared with a two-dose primary series followed by a booster dose of ten-valent or 13-valent pneumococcal conjugate vaccine in South African children: an open-label, randomised, non-inferiority trial -